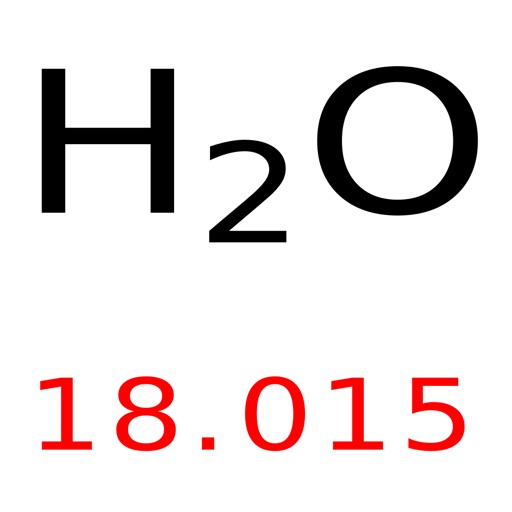 
Performed by New York Philharmonic, conducted by Leonard Bernstein. Robert Millikan - 1923 Nobel Prize in Physicsīorodin, Alexander P. Chicago, IL: University of Chicago Press, 1947. Electrons, Protons, Photons, Neutrons, Mesotrons, and Cosmic Rays. New York, NY: Cornell University Press, 1982. The Rise of Robert Millikan: Portrait of a Life in American Science. New York, NY: Philosophical Library, 1964.Ĭardwell, D. New York, NY: Longmans Green and Co., 1897.ĭalton, John. A Well-Ordered Thing: Dmitrii Mendeleev and the Shadow of the Periodic Table. Mendeleyev’s Dream: The Quest for the Elements. Madison, WI: Journal of Chemical Education, 1968. New York, NY: Oxford University Press, 1998. The lecture ends with a brief biographical sketch of composer and chemistry professor Alexander Borodin, a contemporary of Mendeleev. Sadoway demonstrates how to write a balanced equation, employing stoichiometry to determine how much metal is produced from a given amount of reactants. Using the example of the Kroll process for producing titanium metal, Prof. The lecture includes a description of Robert Millikan’s oil drop experiment (1909), which measured the value of the elementary charge.Ī chemical reaction can be described by an equation based on conservation of mass and Dalton’s law of molar proportions. mole, Avogadro’s number, Faraday’s constant, elementary charge, and atomic mass unit.atomic mass, proton number, neutron number, and isotopes.Starting with the fundamental structure of the atom and characteristics of the electron, proton, and neutron, Prof. Sadoway discusses naming conventions and how these elements are discovered.Įlements are characterized by a range of properties. 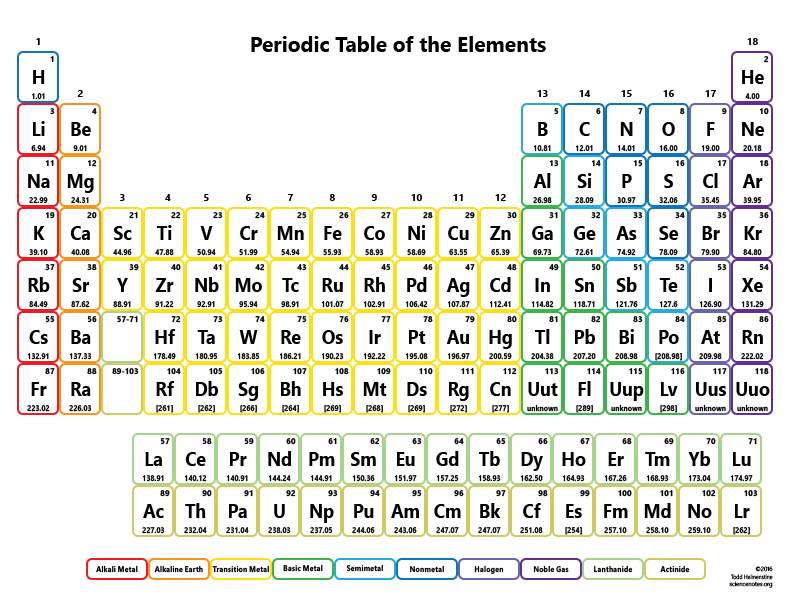
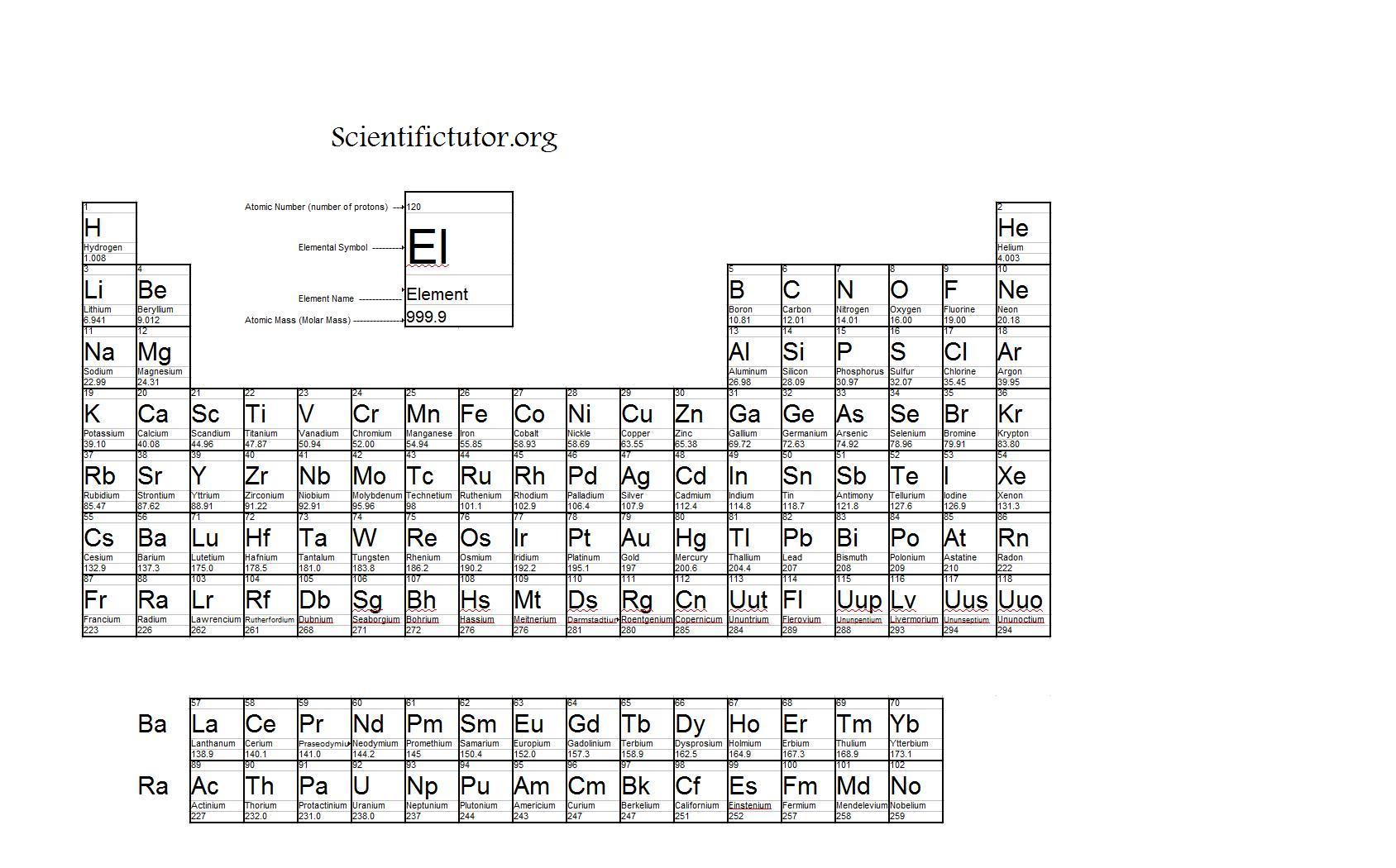
At the high end of the periodic table are the superheavy elements Prof. This lecture continues the discussion about origins of the periodic table, picking up with Dmitri Mendeleev’s discovery of periodic patterns among different groups of elements. Periodic Table and Table of Constants Lecture Summary 7.1, “The History of the Periodic Table.” (only read until “Mendeleev’s Periodic Table”)Īrrangements of elements before Mendeleev Stoichiometry problems limiting reactants percent yields 3.4, “Mass Relationships in Chemical Equations.” Interpreting chemical equations balancing simple chemical equations 3.2, “Determining Empirical and Molecular Formulas.”Ĭalculating mass percentages determining the empirical formula of penicillin combustion analysis from empirical formula to molecular formula Molecular and formula masses the mole molar mass Metals, nonmetals, and semimetals chemistry of the groups 1.7, “Introduction to the Periodic Table.”
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
